 |
||
 |
||
|
|
|
Active Hydrogen
Adrenal Extracts
Alanine
Alpha-Linolenic Acid
Alpha-Lipoic Acid
AMP
Amylase Inhibitors
Arginine
Bee Pollen
Beta Carotene
Beta-glucan
Betaine
Beta-Sitosterol
Biotin
Borage Oil
Boron
Bovine Cartilage
Bovine Colostrum
Brewer's Yeast
Bromelain
Calcium
Capsaicin
Carnitine
Carnosine
Chitosan
Chloride
Chlorophyll
Chondroitin
Chromium
CLA
Cobalt
Coenzyme Q10
Copper
Creatine
Cysteine
DHA
DHEA
DMAE
EGCG
Evening Primrose Oil
5-HTP
Fiber (Insoluble)
Fiber (Soluble)
Fish Oil
Flavonoids
Fluoride
Folate
Fumaric Acid
GABA
Gamma-Linolenic Acid
Glucomannan
Glucosamine
Glutamic Acid
Glutamine
Glutathione
Glycine
Grape Seed Extract
Histidine
HMB
Hydroxycitric Acid
Indole
Inosine
Inositol
Iodine
Ipriflavone
Iron
Isoleucine
Lactase
Lecithin
Leucine
Lipase
Lutein
Lycopene
Lysine
Magnesium
Malic Acid
Manganese
Mannose
Melatonin
Methionine
Methoxyisoflavone
Molybdenum
MSM
N-Acetyl Cysteine
NADH
Naringin
Niacin
Octacosanol
Oligosaccharides
Olive Leaf Extract
Ornithine
Oryzanol
PABA
Pancreatic Enzymes
Pantothenic Acid
Phenylalanine
Phosphatidylserine
Phosphorus
Phytic Acid
Policosanol
Potassium
Pregnenolone
Probiotics
Propolis
Psyllium
Pyridoxine
Pyruvate
Quercetin
Resveratrol
Retinol
Riboflavin
Ribose
Royal Jelly
SAMe
Selenium
Shark Cartilage
Silicon
Sodium
Spirulina
Spleen Extracts
St. John's Wort
Strontium
Sulforaphane
Sulfur
Taurine
Thiamine
Tocopherol
Tea Tree Oil
Tyrosine
Usnic Acid
Valine
Vanadium
Vinpocetine
Vitamin A
Vitamin B1
Vitamin B2
Vitamin B3
Vitamin B5
Vitamin B6
Vitamin B9
Vitamin B12
Vitamin C
Vitamin D
Vitamin H
Vitamin K
Whey Protein
Xylitol
Zinc
Abalone Shell (shi jue ming)
Abutilon Seed (dong kui zi) Acanthopanax Bark (wu jia pi) Achyranthes (niu xi) Aconite (fu zi) Acorus (shi chang pu) Adenophora Root (nan sha shen) Agkistrodon (bai hua she) Agrimony (xian he cao) Ailanthus Bark (chun pi) Akebia Fruit (ba yue zha) Albizzia Bark (he huan pi) Albizzia Flower (he huan hua) Alfalfa (medicago sativa) Alisma (ze xie) Aloe (lu hui) Alum (bai fan) Amber (hu po) Ampelopsis (bai lian) Andrographis (chuan xin lian) Anemarrhena (zhi mu) Antelope's Horn (ling yang jiao) Apricot Seed (xing ren) Areca Peel (da fu pi) Areca Seed (bing lang) Arisaema (tian nan xing) Ark Shell (wa leng zi) Arnebia (zi cao or ying zi cao) Arnica (arnica montana) Artichoke Leaves (Cynara scolymus) Ash bark (qin pi) Ashwagandha (withania somniferum) Aster (zi wan) Astragalus (huang qi) Aurantium (zhi ke [qiao]) Bamboo Juice (zhu li) Bamboo Shavings (zhu ru) Belamcanda Rhizome (she gan) Benincasa Peel (dong gua pi) Benincasa Seed (dong gua xi/ren) Benzoin (an xi xiang) Bilberry (yue ju) Biota Leaf (ce bai ye) Biota Seed (bai zi ren) Bitter Melon (ku gua) Bitter Orange Peel (ju hong) Black Cohosh (sheng ma) Black Plum (wu mei) Black Sesame Seed (hei zhi ma) Bletilla (bai ji) Boneset (ze lan) Borax (peng sha) Borneol (bing pian) Bottle Brush (mu zei) Buddleia (mi meng hua) Buffalo Horn (shui niu jiao) Bulrush (pu huang) Bupleurum (chai hu) Burdock (niu bang zi) Camphor (zhang nao) Capillaris (yin chen hao) Cardamon Seed (sha ren) Carpesium (he shi) Cassia Seed (jue ming zi) Catechu (er cha) Cat's Claw (uncaria tomentosa) Cephalanoplos (xiao ji) Celosia Seed (qing xiang zi) Centipede (wu gong) Chaenomeles Fruit(mu gua) Chalcanthite (dan fan) Chebula Fruit (he zi) Chinese Gall (wu bei zi) Chinese Raspberry (fu pen zi) Chrysanthemum (ju hua) Cibotium (gou ji) Cinnabar (zhu sha) Cinnamon (rou gui or gui zhi) Cistanche (rou cong rong) Citron (xiang yuan) Citrus Peel (chen pi) Clam Shell (hai ge ke/qiao) Clematis (wei ling xian) Cloves (ding xiang) Cnidium Seed (she chuang zi) Codonopsis (dang shen) Coix Seed (yi yi ren) Coptis (huang lian) Cordyceps (dong chong) Coriander (hu sui) Corn Silk (yu mi xu) Cornus (shan zhu yu) Corydalis (yan hu suo) Costus (mu xiang) Cranberry (vaccinium macrocarpon) Cremastra (shan ci gu) Croton Seed (ba dou) Curculigo (xian mao) Cuscuta (tu si zi) Cuttlefish Bone (hai piao xiao) Cymbopogon (xiang mao) Cynanchum (bai qian) Cynomorium (suo yang) Cyperus (xiang fu) Dalbergia (jiang xiang) Damiana (turnera diffusa) Dandelion (pu gong ying) Deer Antler (lu rong) Dendrobium (shi hu) Devil's Claw (harpagophytum procumbens) Dianthus (qu mai) Dichroa Root (chang shan) Dittany Bark (bai xian pi) Dong Quai (tang kuei) Dragon Bone (long gu) Dragon's Blood (xue jie) Drynaria (gu sui bu) Dryopteris (guan zhong) Earthworm (di long) Eclipta (han lian cao) Elder (sambucus nigra or sambucus canadensis) Elsholtzia (xiang ru) Ephedra (ma huang) Epimedium (yin yang huo) Erythrina Bark (hai tong pi) Eucalyptus (eucalyptus globulus) Eucommia Bark (du zhong) Eupatorium (pei lan) Euphorbia Root (gan sui or kan sui) Euryale Seed (qian shi) Evodia (wu zhu yu) Fennel (xiao hui xiang) Fenugreek (hu lu ba) Fermented Soybeans (dan dou chi) Flaxseed (ya ma zi) Fo Ti (he shou wu) Forsythia (lian qiao) Frankincense (ru xiang) Fritillaria (chuan bei mu) Gadfly (meng chong) Galanga (gao liang jiang) Galena (mi tuo seng) Gambir (gou teng) Gardenia (zhi zi) Garlic (da suan) Gastrodia (tian ma) Gecko (ge jie) Gelatin (e jiao) Genkwa (yuan hua) Germinated Barley (mai ya) Ginger (gan [sheng] jiang) Ginkgo Biloba (yin xing yi) Ginseng, American (xi yang shen) Ginseng, Asian (dong yang shen) Ginseng, Siberian (wu jia shen) Glehnia (sha shen) Glorybower (chou wu tong) Goldenseal (bai mao liang) Gotu Kola (luei gong gen) Green Tea (lu cha) Gymnema (gymnema sylvestre) Gynostemma (jiao gu lan) Gypsum (shi gao) Halloysite (chi shi zhi) Hawthorn (shan zha) Hemp Seed (huo ma ren) Homalomena (qian nian jian) Honey (feng mi) Honeysuckle Flower (jin yin hua) Honeysuckle Stem (ren dong teng) Houttuynia (yu xing cao) Huperzia (qian ceng ta) Hyacinth Bean (bai bian dou) Hyssop (huo xiang) Ilex (mao dong qing) Imperata (bai mao gen) Indigo (qing dai) Inula (xuan fu hua) Isatis Leaf (da qing ye) Isatis Root (ban lan gen) Java Brucea (ya dan zi) Jujube (da zao) Juncus (deng xin cao) Kadsura Stem (hai feng teng) Katsumadai Seed (cao dou kou) Kelp (kun bu) Knotweed (bian xu) Knoxia root (hong da ji) Kochia (di fu zi) Lapis (meng shi) Leech (shui zhi) Leechee Nut (li zhi he) Leonorus (yi mu cao) Lepidium Seed (ting li zi) Licorice (gan cao) Ligusticum (chuan xiong) Ligustrum (n zhen zi) Lily Bulb (bai he) Limonite (yu liang shi) Lindera (wu yao) Litsea (bi cheng qie) Lobelia (ban bian lian) Longan (long yan hua [rou]) Lophatherum (dan zhu ye) Loquat Leaf (pi pa ye) Lotus Leaf (he ye) Lotus Node (ou jie) Lotus Seed (lian zi) Lotus Stamen (lian xu) Luffa (si gua luo) Lycium Bark (di gu pi) Lycium Fruit (gou qi zi) Lygodium (hai jin sha) Lysimachia (jin qian cao) Magnetite (ci shi) Magnolia Bark (hou po) Magnolia Flower (xin yi hua) Maitake (grifola frondosa) Marigold (c. officinalis) Massa Fermentata (shen qu) Milk Thistle (silybum marianum) Millettia (ji xue teng) Mint (bo he) Mirabilite (mang xiao) Morinda Root (ba ji tian) Mugwort Leaf (ai ye) Mulberry Bark (sang bai pi) Mulberry Leaf (sang ye) Mulberry Twig (sang zhi) Mullein (jia yan ye) Musk (she xiang) Myrrh (mo yao) Notoginseng (san qi) Notopterygium (qiang huo) Nutmeg (rou dou kou) Oldenlandia (bai hua she she cao) Omphalia (lei wan) Onion (yang cong) Ophicalcite (hua rui shi) Ophiopogon (mai dong) Oroxylum Seed (mu hu die) Oryza (gu ya) Oyster Shell (mu li) Passion Flower (passiflora incarnata) Patrinia (bai jiang cao) Pau D'Arco (tabebuia avellanedae) Peach Seed (tao ren) Pearl (zhen zhu [mu]) Perilla Leaf (su ye) Perilla Seed (su zi) Perilla Stem (su geng) Persimmon (shi di) Pharbitis Seed (qian niu zi) Phaseolus (chi xiao dou) Phellodendron (huang bai) Phragmites (lu gen) Picrorhiza (hu huang lian) Pinellia (ban xia) Pine Knots (song jie) Pipe Fish (hai long) Plantain Seed (che qian zi) Platycodon (jie geng) Polygala (yuan zhi) Polygonatum (huang jing) Polyporus (zhu ling) Poppy Capsule (ying su qiao) Poria (fu ling) Prickly Ash Peel (hua jiao) Prinsepia Seed (rui ren/zi) Prunella (xia ku cao) Prunus Seed (yu li ren) Pseudostellaria (tai zi shen) Psoralea (bu gu zhi) Pueraria (ge gen) Pulsatilla (bai tou weng) Pumice (fu hai shi) Pumpkin Seed (nan gua zi) Purslane (ma chi xian) Pyrite (zi ran tong) Pyrrosia Leaf (shi wei) Quisqualis (shi jun zi) Radish (lai fu zi) Realgar (xiong huang) Red Atractylodes (cang zhu) Red Clover (trifolium pratense) Red Ochre (dai zhe shi) Red Peony (chi shao) Red Sage Root (dan shen) Rehmannia (shu di huang) Reishi (ling zhi) Rhubarb (da huang) Rice Paper Pith (tong cao) Rose (mei gui hua) Rosemary (mi die xiang) Safflower (hong hua) Saffron (fan hong hua) Sandalwood (tan xiang) Sanguisorba Root (di yu) Sappan Wood (su mu) Sargent Gloryvine (hong teng) Saw Palmetto (ju zong lu) Schefflera (qi ye lian) Schisandra (wu wei zi) Schizonepeta (jing jie) Scirpus (san leng) Scopolia (S. carniolica Jacq.) Scorpion (quan xie) Scrophularia (xuan shen) Scutellaria (huang qin) Sea Cucumber (hai shen) Sea Horse (hai ma) Seaweed (hai zao) Selaginella (shi shang bai) Senna (fan xie ye) Shiitake (hua gu) Siegesbeckia (xi xian cao) Siler Root (fang feng) Slippery Elm (ulmus fulva) Smilax (tu fu ling) Smithsonite (lu gan shi) Sophora Flower (huai hua mi) Sophora Root (ku shen) Spirodela (fu ping) Stellaria (yin chai hu) Stemona (bai bu) Stephania (fang ji [han]) Sweet Annie (qing hao) Teasel Root (xu duan) Tiger Bone (hu gu) Torreya Seed (fei zi) Tortoise Plastron (gui ban) Tremella (bai mu er) Trichosanthes Fruit (gua lou) Trichosanthes Root (tian hua fen) Trichosanthes Seed (gua lou ren) Tsaoko Fruit (cao guo) Turmeric (jiang huang) Turtle Shell (bie jia) Tussilago (kuan dong hua) Urtica (xun ma) Uva ursi (arctostaphylos uva-ursi) Vaccaria Seed (wang bu lui xing) Valerian (jie cao) Veratrum (li lu) Viola (zi hua di ding) Vitex (man jing zi) Walnut (hu tao ren) Watermelon (xi gua) White Atractylodes (bai zhu) White Mustard Seed (bai jie ze) White Peony (bai shao) Wild Asparagus (tian men dong) Windmill Palm (zong lu pi/tan) Xanthium (cang er zi) Zedoary (e zhu) |
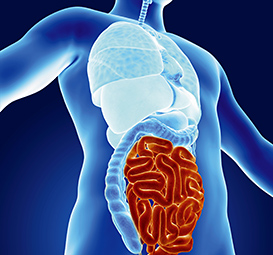
Your Gut: Why You Should Be Very Afraid
Protect your intestinal flora before it's systematically depleted.
By Marlene Merritt, LAc, DOM We think it's so easy — just take that probiotic to make up for whatever damage might have happened from the prescription antibiotic your doctor gave you, and you'll be fine, right? And that antibiotic — you hear about antibiotic resistance, but you take it correctly, and you really needed it for that sinus infection, so it's not that big of a deal either, right? You're wrong in ways you can't possibly imagine – and that should make you very, very nervous. The World Health Organization has started to use the phrase, "The Post-Antibiotic Era," to describe our current period of time. In September 2013, the Centers for Disease Control and Prevention issued its first solid numbers regarding antibiotic resistance. It estimates at least 2 million Americans fall ill to antibiotic-resistant infections and between 23,000 to 100,000 people die from them every year. There are 23 known bacteria that are completely resistant to anything we throw at them and that number, of course, is climbing. This resistance has occurred for a couple of reasons. First, the CDC says 50 percent of the prescriptions written for antibiotics are unnecessary or inappropriate (for example, a broad-spectrum antibiotic is given when a culture isn't taken to determine a more targeted drug). Second, 80 percent of available antibiotics are actually given to animals in confined feed lots (which might make you decide to buy grass-fed, or at least, antibiotic-free, meat). Roundup® glyphosphate, found in conventionally raised farming, has been shown to have an inhibitory effect on food microorganisms and suppress beneficial bacteria in the gut.
Think about all the times in your life you've taken an antibiotic — it's most often for respiratory conditions and yet is the least effective. The person who says, one or two months after their sinus infection, that they've gotten another one, probably has the same infection that wasn't properly handled or was resistant to medication. When people feel better taking an antibiotic, it's often just because of a reduction in the pathogenic bacteria, not because it's actually killing them off; and it's been shown time and time again that taking an antibiotic only reduces your illness time by a day or two. "But that's a lot!" you cry. However, if you knew taking that antibiotic might permanently damage your gut flora or cause other bacteria to become resistant, you might pause or possibly decline taking that medicine. And it's now been found that bacteria "talk" to each other, and convey resistance to medications. Yes, that means bacteria are passing on antibiotic resistance to other bacteria. The biggest problem is that our gut flora can't take this abuse. Let's start with some basics that will explain why this is potentially such a problem. Impact on Gut Flora You inherit your gut flora from your mother in a vaginal birth and continue to populate your gut with different strains from the air, food, soil, etc., until the age of 2, when it's "set." Going back to your mother, how healthy was her gut flora? How many antibiotics did she take in her lifetime? How about your grandmother? We've had antibiotics for 80 years now and while there's no doubt they've saved a lot of lives, there's also no doubt they are less and less effective, and the collective damage to gut flora is irreparable. Add in the high Caesarean section rate and you can begin to get an idea that our gut flora are currently the weakest in human history. Studies are showing that babies born by C-section have higher rates of celiac disease and autoimmune disorders like type 1 diabetes and obesity. Our gut flora outnumber our body cells by 10-to-1. Yes, we're only 10 percent human. The Human Microbiome Project is attempting to distinguish all the different actions and functions of the gut flora, but to give you some idea: It is estimated that 70-90 percent of your immune system resides in your gut. Autoimmune diseases have been directly linked to imbalances in gut flora, and celiac disease has been directly linked to dysbiosis. I dealt with a patient in her 60s who got debilitating celiac disease after a single round of antibiotics, and another who improved his ulcerative colitis 80 percent by not washing the vegetables from his garden. Gut bacteria are responsible for B vitamins, vitamin K, and the production of amino acids and fatty acids. It is now thought that gut microbes, along with some of the inflammatory foods we're eating, like junk food and bad fats, make endotoxins, which are one of the root causes of inflammation. Rats with no gut microbes can eat any amount of "bad" foods and not gain weight or have inflammation, but when given gut flora, immediately develop high levels of endotoxins and the resulting inflammation. And those endotoxins? Huge contributors to leaky gut syndrome. Your gut has the same number of neurons as your spinal cord, and about 95 percent of your body's serotonin is produced in the gut, as well as many other neurotransmitters. It almost seems odd, then, to have prescriptions for SSRI medications and never actually deal with what might be happening in the gut. Your gut flora individuality is like a fingerprint, in that it's different for everyone. That being said, diversity is the key to health in regards to microbes; and if you don't have diversity, or you don't have the right type of bacteria, it leaves you open to a host of diseases, including obesity. It's this diversity of your gut, not just the number of bacteria, which seems to be key to staying healthy. First World children have less diversity than Third World children, with corresponding levels of asthma and allergies, and even though rural Third World countries have greater exposure to infectious diseases and lower life expectancies, they also have much lower rates of overall degenerative and chronic diseases. Irreparable Damage? The use of antibiotics is what's frightening — they don't just deplete gut bacteria, but can actually permanently wipe out whole strains. Scientists are now going into remote areas of the Amazon and collecting gut bacteria from tribes that have never been exposed to Westerners, antibiotics or modern foods, and are finding that a pristine microbiome has huge diversity, including species never before sequenced. Of course, how we have tended to look at bacteria is a "good" versus "bad" scenario, but it is much more complicated than that. First, everyone has pathogenic bacteria in their gut, but they are often kept under control by adequate amounts of beneficial bacteria. Take an antibiotic, however, wiping down or out our good guys, and the bad guys can flourish, leading to deadly Clostridium difficile infections, for example. Or take Helicobacter pylori, which we've been trying to exterminate since 1983, when it was linked to peptic ulcers. But it's also been shown that H. pylori regulates acid in the stomach — when people eliminate their H. pylori, they have less peptic ulcers, but higher rates of acid reflux, Barrett's esophagus, and esophageal cancer, rates of which have soared since we've been eliminating H. pylori. It also calms the immune system (populations with higher H. pylori have lower rates of allergy and asthma), and regulates ghrelin and correspondingly, metabolism; obesity related to low H. pylori levels is currently being investigated. What happens if you wipe out your home team so badly it can't recover? The symptoms can range from chronic, debilitating diarrhea, to malnutrition, anemia, systemic infections and death. When faced with a pathogenic infection as a result of taking antibiotics, most doctors employ the very method that got the patient in trouble in the first place — antibiotics — but what is becoming a quickly developing (and more effective) treatment is fecal transplants. A fecal transplant takes the gut bacteria from a healthy person and introduces it to the sick person, often with shockingly dramatic improvements. In a study done with patients who had a C. difficile infection and never properly recovered (most had more than 150 days of incapacitating diarrhea), a fecal transplant reversed their symptoms in 24 hours, on average, with a total cure rate of over 90 percent. In fact, they halted the study because it became unethical to withhold such a successful treatment from the control group. The problem is, you can't actually replace what you've lost. We're under the mistaken impression that you can simply take an antibiotic and then follow with a probiotic, and your gut will be completely back to normal – but that's not what being seen in research. Even fecal transplants, with their dramatic impacts, simply buy time for the home team to recover. Probiotics also help to keep the "bad guys" in check, but don't replace lost strains. If my system got "flushed out" with some food-poisoning diarrhea, for example, I might use a probiotic pill to help my gut out while it repopulates, but when someone has to rely on probiotic pills for their digestion to be "normal," that's an indication that there's some dysbiosis / dysfunction in their gut. Taking higher and higher amounts of probiotics isn't great, either — buying one with insane amounts of colony-forming units (CFUs) can cause dysbiosis as well. And of course, there's no probiotic specific for your individual microorganism makeup. A Preservation Strategy Prebiotics, like the oligosaccharides found in vegetables and whole grains, as well as in breast milk (to encourage gut flora growth in a baby), are like food and housing for your home team — they encourage your native flora to grow. Focusing on good prebiotics, as well as severely reducing foods like sugar and bad fats (which cause that inflammatory reaction with the gut flora and can encourage dysbiosis) would be a better plan of action. "What if I take my home team and cultivate them in a Petri dish for sometime in the future?" I sometimes get asked. That doesn't work, either — when implanted back into the gut, your original "home team" bacteria don't survive long enough to reinoculate. This is where the impact starts to hit people — when we begin to realize that we're destroying the biggest and most complicated "organ" we have from sheer lack of understanding. In labeling bacteria "good" and "bad," we've sanitized our houses, our skin, our bodies and our kids. The result is a confused and weakened immune system, obesity, neurological dysfunction, chronic diseases, and more modern, deadly diseases. It was after all this research that I began to realize I would literally have to be dying to take another antibiotic. It makes more sense to keep my immune system strong, and use herbs and nutrition to fight infections if necessary, while in the meantime regularly eating the traditional foods that gave us a little probiotic help — fermented foods like sauerkraut, kimchi, kombucha, lacto-fermented vegetables, etc. It's not just the specter of antibiotic resistance that should scare people silly. It's also the depletion of their own gut flora and the resulting physiological damage for following generations that might make you, or your now-educated patients, choose differently the next time they're in the doctor's office. Resources
Marlene Merritt is a licensed acupuncturist and runs a wellness center in Austin, Texas. She specializes in combining Oriental medicine, nutrition and Western testing to maximize patient care. Contact her with questions and comments by clicking here. |
Nutritional Wellness News Update:

Other Alternative Health Sites
Toyour Health
ChiroWeb
ChiroFind
Dynamic Chiropractic
DC Practice Insights
Acupuncture Today
Massage Today
Naturopathy Digest
Chiropractic Research Review
Spa Therapy
|


 You probably have no idea (and most patients certainly are in the dark) how ineffective antibiotics are for the things they're prescribed for most often. They're not effective for bronchitis, sinus infections or ear infections. They are incorrectly prescribed in 80 percent of strep infections. And of course, they are often given for the common cold or the flu, despite the fact that both are viral, because either the patient demands a prescription or the doctor wants to prophylactically prevent a secondary bacterial infection.
You probably have no idea (and most patients certainly are in the dark) how ineffective antibiotics are for the things they're prescribed for most often. They're not effective for bronchitis, sinus infections or ear infections. They are incorrectly prescribed in 80 percent of strep infections. And of course, they are often given for the common cold or the flu, despite the fact that both are viral, because either the patient demands a prescription or the doctor wants to prophylactically prevent a secondary bacterial infection.